 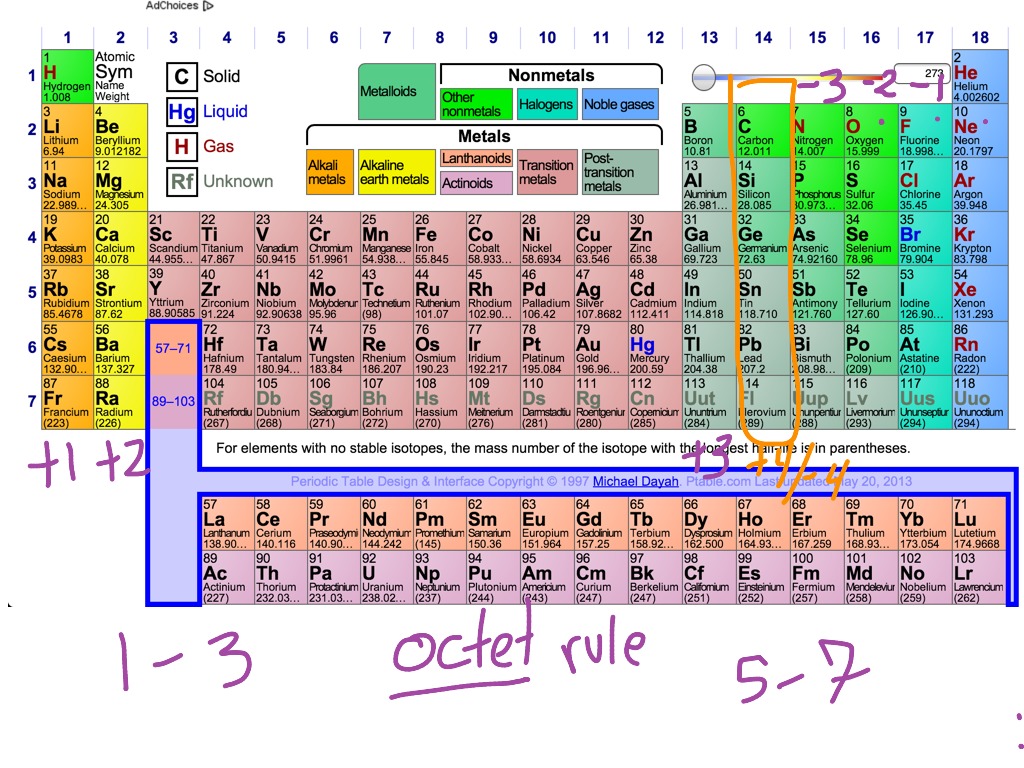
This is the reason why H is always a terminal atom and never a central atom. The electron dots in the Lewis structure are a convenient way to determine how many bonds an atom of an element can make. 1: Lewis symbols or electron-dot symbols of the first twenty elements in the periodic table. aluminum oxide, Al 2 O 3: aluminum has a valence of 3. Across a row of the periodic table, generally Z eff of the valence electrons increases across a period, but the trend does depend on shell and subshell. hydrogen sulfide, H 2 S: sulfur has a valence of 2. This trend is best illustrated by inspection of Figure 1.17.1 1.17. Hydrogen only needs to form one bond to complete a duet of electrons. Helium is an exception that has only two valence electrons, but they are shown paired. lithium oxide, Li 2 O: lithium has a valence of 1. Study with Quizlet and memorize flashcards containing terms like The number of moles of Cl - ions needed to combine completely with 0. Atom (Group number)īecause hydrogen only needs two electrons to fill its valence shell, it follows the duet rule. Table showing 4 different atoms, each of their number of bonds, and each of their number of lone pairs. In each case, the sum of the number of bonds and the number of lone pairs is 4, which is equivalent to eight (octet) electrons. The number of electrons required to obtain an octet determines the number of covalent bonds an atom can form. 
Oxygen and other atoms in group 16 obtain an octet by forming two covalent bonds: To obtain an octet, these atoms form three covalent bonds, as in NH 3 (ammonia). Group 15 elements such as nitrogen have five valence electrons in the atomic Lewis symbol: one lone pair and three unpaired electrons. The transition elements and inner transition elements also do not follow the octet rule since they have d and f electrons involved in their valence shells. Valence electrons: For main group elements (i. A Lewis symbol consists of an elemental symbol surrounded by one dot for each of its valence electrons: Figure 7.9 shows the Lewis symbols for the elements of the third period of the periodic table. Because hydrogen only needs two electrons to fill its valence shell, it is an exception to the octet rule and only needs to form one bond. We use Lewis symbols to describe valence electron configurations of atoms and monatomic ions. These four electrons can be gained by forming four covalent bonds, as illustrated here for carbon in CCl 4 (carbon tetrachloride) and silicon in SiH 4 (silane). Using the variable n to represent the number of the valence electron shell, write the valence shell electron configuration for each group. For example, each atom of a group 14 element has four electrons in its outermost shell and therefore requires four more electrons to reach an octet. The shape of the periodic table reflects the order in which electron shells and subshells fill with electrons. The number of bonds that an atom can form can often be predicted from the number of electrons needed to reach an octet (eight valence electrons) this is especially true of the nonmetals of the second period of the periodic table (C, N, O, and F). From left to right: water molecule, ammonia molecule, and methane molecule 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
